Barrier rigid films
For pharmaceuticals that require more stringent moisture and oxygen protection, these highly protective films ensure a long shelf life with optimal product integrity. kp offers a wide range of ACLAR® laminated and alfoil® PVdC coated barrier films for your specific application.
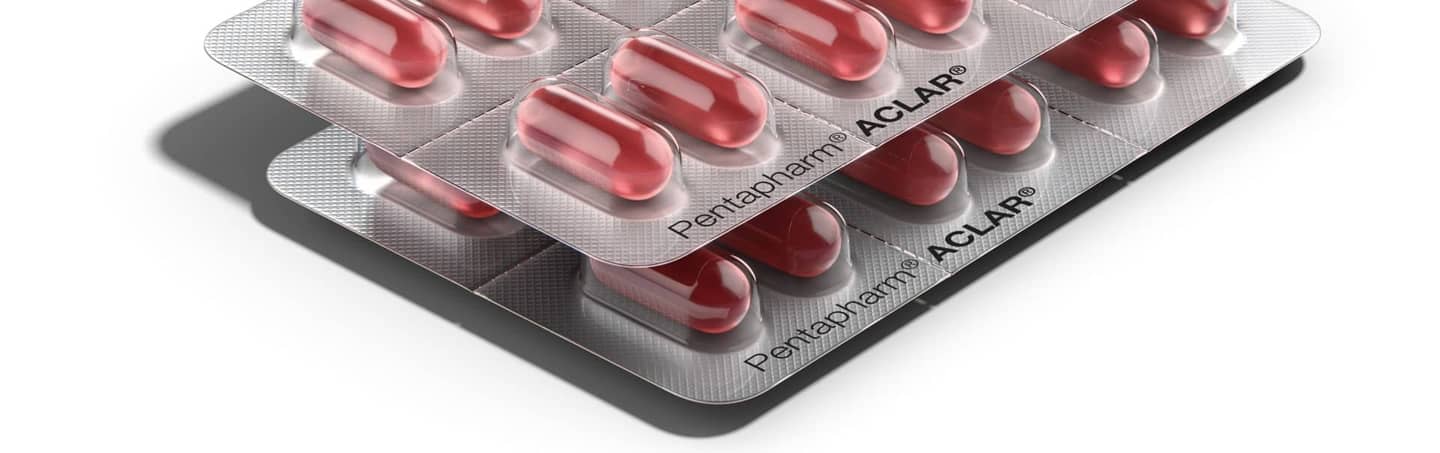
Pentapharm® ACLAR® laminated films
Pentapharm® ACLAR® laminates are crystal clear, biochemically inert, thermally stable with unsurpassed protection against moisture in formulations designed to significantly improve performance and productivity. In addition to ultra-high moisture barrier, Pentapharm® ACLAR® laminates are available in multi-layer structures with oxygen protection.
Applications:
Ethicals, over the counter (OTC), generics, physician samples, veterinary, dietary supplements and nutraceuticalsBenefits:
- Best-in-class moisture barrier
- Non-aging for lasting optical clarity
- Full range of barrier protection
- Possible 40% reduction in surface area of the blister allowing up to 30% higher machine output
- Runs on standard equipment and tooling
- Excellent sealability
- Available in clear, transparent, and opaque colors with UV-absorber on request
Structure:
- Two-layer, three-layer and symmetrical structures standard
- Customized structures available on request
ACLAR®
is a registered trademark of Honeywell International Inc.
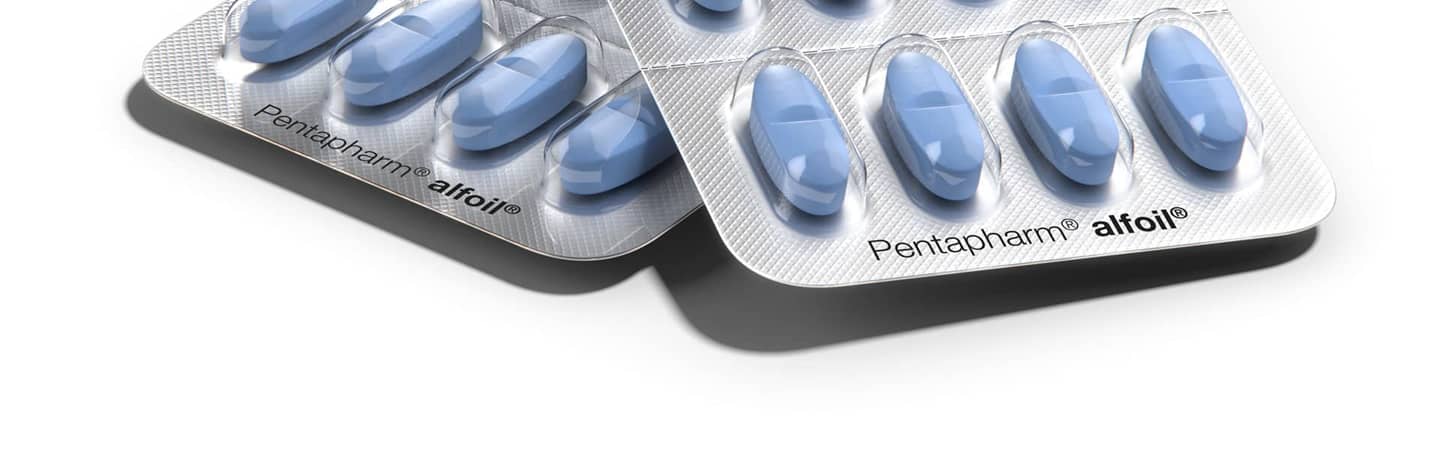
Pentapharm® alfoil® PVdC coated films
Pentapharm® alfoil® is a high-performance line of PVdC-coated films that provide mid-to-high barrier to moisture, oxygen and aroma. Available in a wide range of coat weights and structures, alfoil® films are a cost-effective solution for most hygroscopic and oxygen-sensitive pharmaceuticals. Also available with kp’s proprietary Super Glide (SG) coating technology, alfoil® SG films have improved slip-characteristics that enable faster line speeds, higher yields and lower overall packaging costs.
Applications:
Ethicals, over the counter (OTC), generics, physician samples, veterinary, dietary supplements and nutraceuticalsBenefits:
- Exceptional strength and dimensional stability
- Controlled shrinkage
- Greater rigidity and lay-flat properties for easier cartoning, handling and enhanced aesthetics
- Excellent sealability
- SG developments for higher line speeds and lower costs
- Available in clear, transparent and opaque colors with UV-absorber on request
Structure:
- Two-layer, three-layer, and symmetrical structures standard
- Customized structures available on request
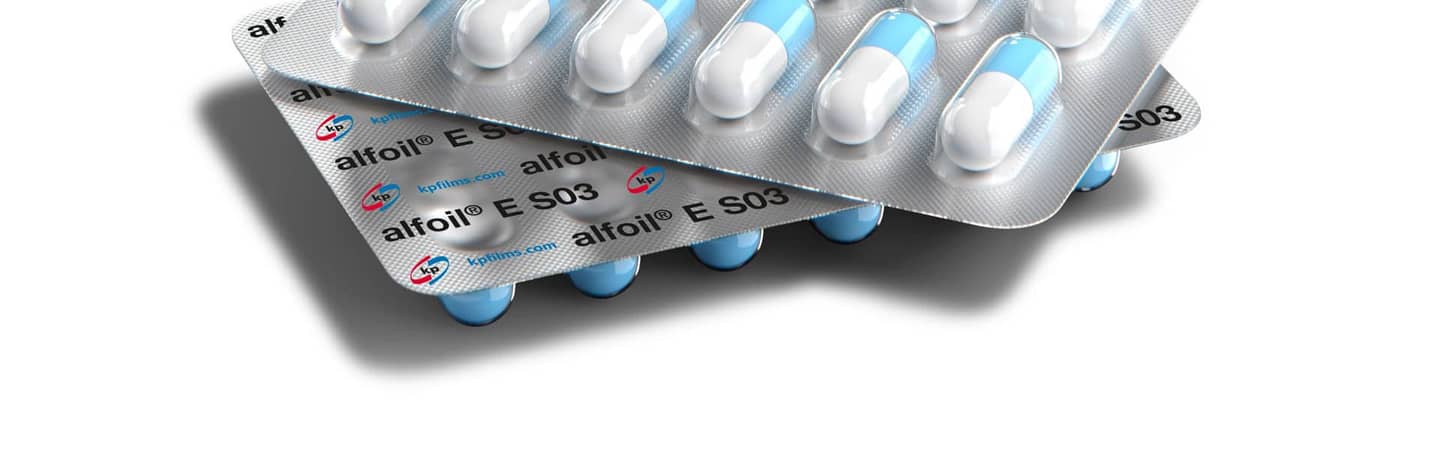
Pentapharm® alfoil® HBC films
Engineered with the latest in ultra-high barrier PVdC technology, alfoil® high-barrier coated (HBC) films offer unrivaled protection against moisture, oxygen and aroma.
Applications:
Ethicals, over the counter (OTC), generics, probiotics, dietary supplements and nutraceuticalsBenefits:
- Ultra-high degree of barrier to moisture, oxygen and aroma
- High-crystallinity PVdC dispersion allows for higher barrier levels at competitive cost
- Excellent thermoforming properties and mechanical stability compared to standard PVdC-coated films
- SG developments for higher line speeds and lower costs
- Brilliant clarity for transparent blisters
- Available in clear, transparent and opaque colors with UV-absorber on request
Structure:
- Three-layer and symmetrical structures standard
- Customized structures available upon request
These laminates combine PVC with PCTFE (Aclar), creating films that are optically clear, chemically inert, and dimensionally stable. Available in duplex or triplex formats such as PVC PCTFE or PVC PCTFE PVC, they offer ultra-low WVTR and preserve clarity over time.
Alfoil films use a PVC base with precision-coated PVDC layers to deliver strong resistance to moisture, oxygen, and aroma. Multiple coating options from 40 to 180 gsm allow custom barrier profiles. kp’s Super Glide option enhances processing speed and yield.
HBC films utilize high-crystallinity PVDC to achieve maximum barrier performance. Ideal for highly sensitive drugs, these films minimize permeability and support extended shelf life.
Barrier Rigid Films Overview
Barrier rigid films are multilayered structures engineered to shield pharmaceuticals from moisture and oxygen ingress. These materials ensure long-term protection and stability of sensitive active ingredients.
Materials and Structure
These films feature a rigid PVC core, laminated with high-barrier materials such as PCTFE (Aclar) or coated with PVDC (alfoil). Duplex and triplex constructions such as PVC PVDC or PVC PCTFE offer tailored performance, with typical base gauges of 250 to 300 microns and coatings between 40 to 180 gsm.
Barrier Performance
- Uncoated PVC: WVTR around 3.0 grams per square meter per day and OTR around 20 cc per square meter per day
- PVDC-coated films: WVTR approximately 0.25 to 0.65; OTR approximately 0.1 to 1
- PCTFE laminates: Extremely low WVTR between 2 and 6
- Some structures such as kpMax maintain less than 5 percent moisture gain in accelerated aging
Regulatory Compliance
All Pentapharm films meet international pharma packaging regulations. They comply with:
- Ph. Eur. 3.1.11 and USP 661
- FDA 21 CFR, EU 10 2011, and JP Food Contact
- ISO 15378 and ISO 9001 2015
Applications
These films are used in blister packs for sensitive products such as antibiotics, probiotics, OTC drugs, and supplements. Options for tinted, transparent, or UV-filtered versions are available.







